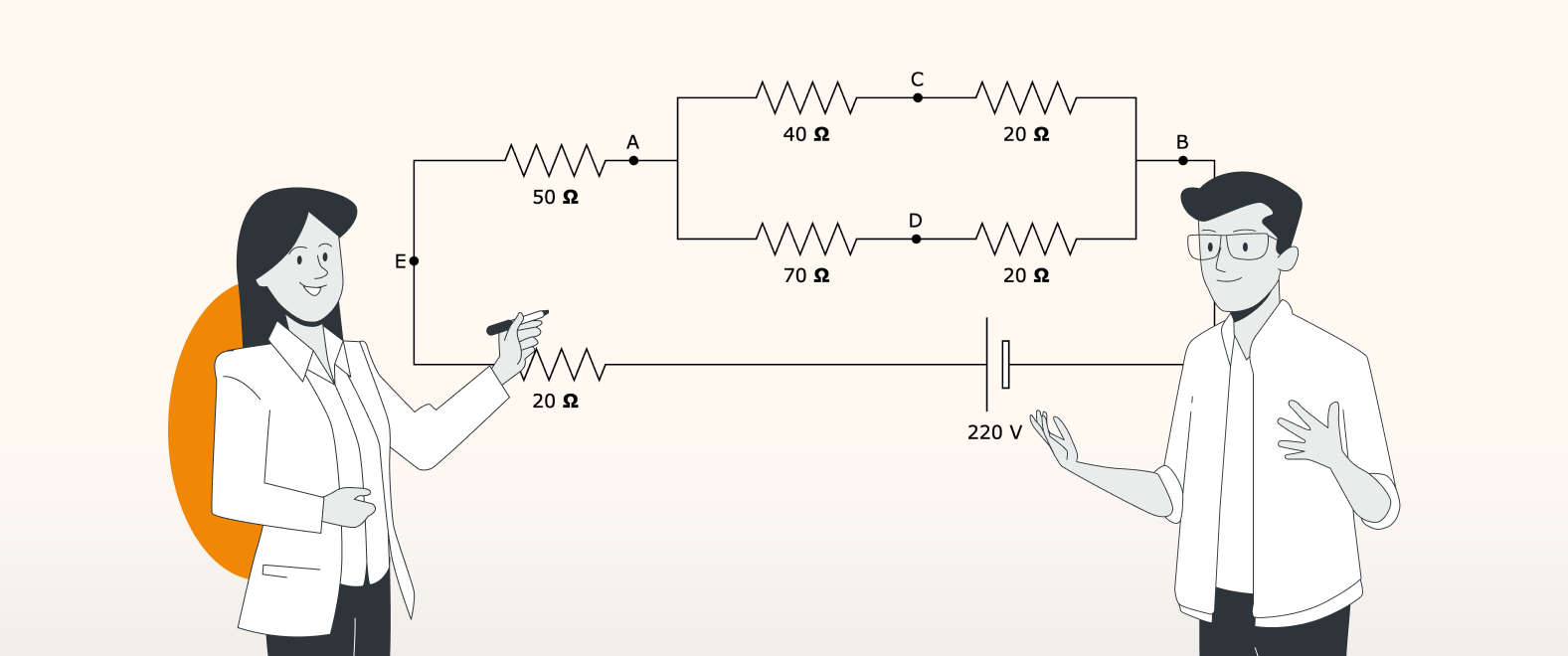
How draw.io and SoftComply help Confluence teams build an ISO-compliant QMS
The gold standard The world of medical device compliance is a tricky one to navigate. At the top level, you have regulations from the FDA in the US, and the MDR/IVDR in Europe. Broken down, you’re then looking at sector-specific requirements like 21 CFR Part 820 in the US, and ISO 13485 + ISO [...]